Yonehara group discovers a new function of a specific type of retinal nerve cells and uncovers a nerve network that transmits visual information from retinal nerve cells into the cerebral cortex.
The new study, published this week in Nature Communications, was led by the neural circuitry team at DANDRITE. The researchers’ findings are important for understanding the mechanisms on how visual perceptions arise within the brain. In this article, first author of the paper, PhD-student at DANDRITE Rune Nguyen Rasmussen, sheds a light on their new findings and its significance for understanding the visual system.
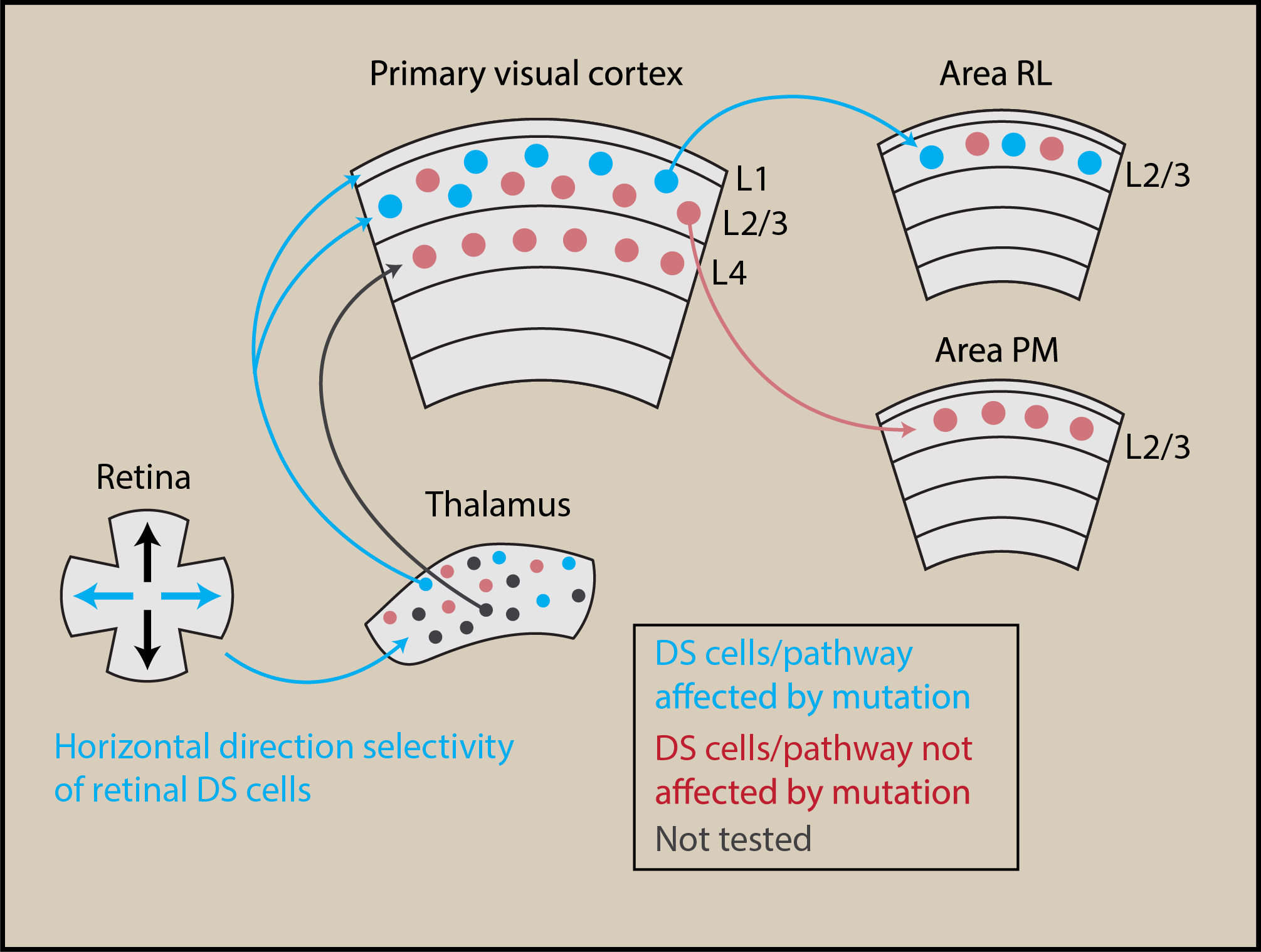
The brain is unquestionably the most complicated and mysterious organ of the body, and our mechanistic understanding of how the brain achieves all of its impressive and vital functions is still in its infancy. The research carried out in this study is essential basic science research, important because it vastly expands our knowledge on how the brain senses, processes and represents sensory information from our surroundings. In this work, we discovered a yet unknown function of a distinct population of nerve cells located within the retina of the eye. We uncovered a specialized nerve network that propagates visual motion information from the nerve cells in the retina and high up into the cerebral cortex. Mapping and understanding the networks within the brain that sends information about specific sensory features is important in order for us to begin to understand how perceptions emerge within the brain at a mechanistic level.
Our work provides several new and significant insights. It had been known for several decades that the retina contains nerve cells sensing the direction of motion when a visual object is moving (so called direction-selective cells). However, the contribution of these cells to the properties of nerve cells in higher-order brain areas of the cerebral cortex was not known, and neither was the pathway linking these two types of nerve cells. By combining several advanced experimental methodologies and a congenital disease mouse model, in which motion processing in the retina is impaired, we are able to provide answers to both of these questions. We showed that retinal nerve cells provide nerve cells of the cerebral cortex with the ability to signal visual motion moving at high velocities; indicating that what has been believed as an important function of cerebral cortex is actually already implemented at the earliest stage of visual processing: the retina. These results thus urge us to update our fundamental model of the physiology of the visual system. Furthermore, we were able to document the detailed pathway that propagates visual motion information from the retina and into distinct higher-order areas of the cerebral cortex.
Our work is of broad interest to all scientists aiming to understand brain functions at a mechanistic and causal level. In particular, these results will be interesting for researchers studying how the brain senses and processes sensory information and for medical doctors who investigate the causes of impaired visual functions. Our work may provide fundamental insights into the mechanisms governing how sensory organs convey information from the periphery to the nerve cells of the cerebral cortex, ultimately giving rise to our perceptions in the world, such as vision, smell and audition. An important remaining question from our work is to determine how and when this nerve pathway is involved in various different behaviors. Thus, in the near future, we will initialize a research project wherein we aim to determine if our identified pathway contribute to the creation of the internal sense of self movements, and spatial navigation. Furthermore, we wish to understand how the functioning of this nerve pathway may be impaired in neurological diseases such as congenital nystagmus or Williams-Beuren syndrome, in which the ability to process and perceive visual motion is impaired in the retina or cerebral cortex, respectively.
The publication can be read in full on the Nature Communications Website.
Click here to learn more about the Yonehara group and their research within structure, function and development of neural circuits in the visual system.