Koning, Jasper T.; Bollmann, Ulla E.; Bester, K
Journal of Hazardous Materials, 406, 124755, 2021.
doi.org/10.1016/j.jhazmat.2020.124755
Abstract:
The environmental fate for some selected antifouling biocides, dichlofluanid, tolylfluanid, tralopyril, and medetomidine, is relatively poorly understood with nearly all data derived from the assessment reports. Water/sediment systems and biofilms were used to determine biodegradation of the antifouling biocides. Dichlofluanid and tolylfluanid are known to hydrolyze to form DMSA (N,N-dimethyl-N′-phenylsulfamide) and DMST (N,N-Dimethyl-N′-(4-methylphenyl)sulfamide), respectively. DMSA did not show biodegradation, but it was shown to transform abiotically into N,N-dimethylsulfamide (N,N-DMS). In contrast, the structurally similar DMST did show biodegradation with a half-life of 5.78 days. The resulting transformation product of the biodegradation of DMST is also N,N-DMS. N,N-DMS accounted for the majority of the mass balance after 27 days in the water/sediment systems. Moreover, the biofilm systems also degraded both DMSA and DMST to N,N-DMS. The hydrolysis product of tralopyril, called BCCPCA (3-bromo-5-(4-chlorophenyl)−4-cyano-1 H-pyrrole-3-carboxylic acid), was not metabolized in the experiments and remained persistent. For this compound, a new log Kow of 2.47 was determined since the previously reported Kow value seemed to overestimate sediment partitioning. Medetomidine was removed from the water/sediment system, though, not significantly more than the control. However, a transformation product (medetomidine-acid) was detected in the incubation but not in the control, pointing to limited biodegradation. These results show that tolylfluanid can be rapidly removed by biodegradation in the marine environment, while dichlofluanid, tralopyril, and medetomidine remained in the system for a longer period of time. The prolonged stability of these biocides could mean that there is potential for accumulation in the environment. This potential is also there for the DMSA (dichlofluanid) and DMST (tolylfluanid) derived transformation product N,N-DMS, which was recalcitrant.
Graphical abstract:
Structural formulas:
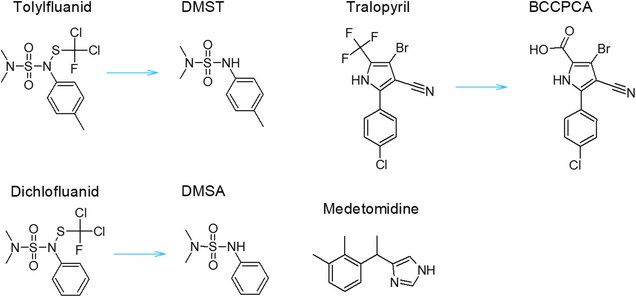
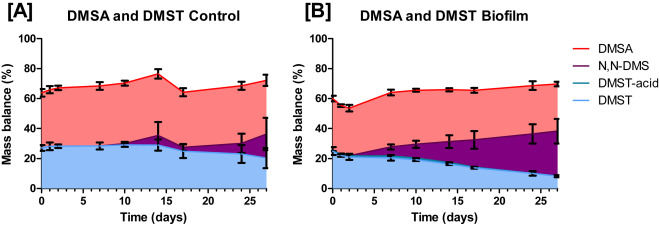
Fig. 4. Biodegradation of DMSA and DMST in biofilms. The y-axis represents the mass balance (in %) that the compound encompasses. The x-axis represents time in days. Filled in areas indicate the share of the specific biocide in the mass balance. All data is plotted as means ± standard deviation (n = 3).